-
Nu.Q® Technology
Our technology detects characteristic epigenetic changes in nucleosomes that occur from the earliest stages of cancer, sepsis and other diseases.
-
- Our Tests
- Human Health
-
Nu.Q® NETs
Nu.Q® NETs is a groundbreaking CE-marked diagnostic solution that clinicians can use to detect NETosis.
-
Nu.Q® Discover
Buy our Nu.Q® Discover H3.1 Research Use Only Assay
- Animal Health
-
Nu.Q® Vet Cancer Test
Nu.Q® Vet Cancer Test is an affordable, accessible blood test that detects cancer in dogs.
-
- Our Pipeline Development
-
Nu.Q® Cancer
We are developing a range of blood-based assays with potential applications beyond cancer detection.
-
Capture-Seq™
We have developed Capture-Seq™, a novel liquid biopsy method designed to isolate and analyze pure circulating tumor-derived DNA (ctDNA) from blood samples.
Volition Announces Submission of Manuscript for Nu.Q® Vet Cancer Test in Cats. The publication of this study in a peer reviewed journal is expected subsequently to unlock a $5 million contractual milestone payment
- Share
- Tweet
- Share on Facebook
- Share
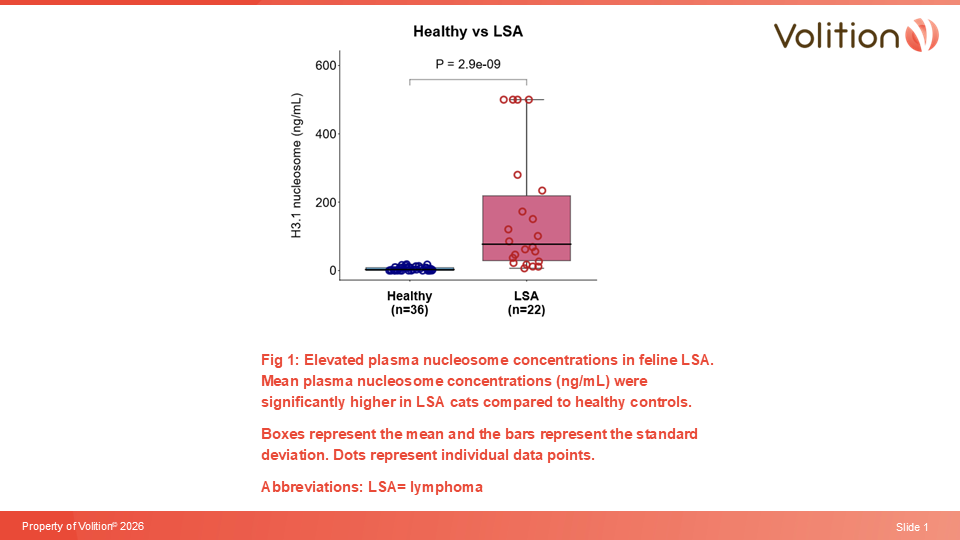
Volition announces submission for peer review a clinical manuscript1 reporting the high accuracy of its Nu.Q® Vet Feline prototype assay in detecting lymphoma in cats, the most common cancer in the species2. At 97% specificity the assay detected 86% of feline lymphomas1 . This breakthrough marks the development of what we expect to be the world’s first simple, affordable blood-based liquid biopsy test for feline cancer, a significant unmet need in veterinary medicine.
Dr Annalisa Canale, Volition and first author on the paper commented:
“Feline oncology lacks tumor biomarkers and so cancer in cats have been notoriously difficult to diagnose early.
“Building on our prior findings for canine lymphoma3-5, this study aimed to highlight the utility of circulating nucleosomes as biomarker to differentiate healthy vs intermediate-large cell lymphoma in cats.
“Nucleosome levels were significantly higher in cats with intermediate-large cell lymphoma compared to healthy controls indicating the potential role of H3.1-nucleosomes as a new useful biomarker for screening intermediate-large cell lymphoma in cats.”
Gael Forterre, Chief Commercial Officer, Volition commented:
“This represents a tremendous commercial opportunity for Volition:
- the publication of this study in a peer reviewed journal is expected subsequently to unlock a $5 million contractual milestone payment; and
- we plan to generate ongoing revenue in this large and growing market where our technology meets an unmet need.
“Our mission is to make cancer screening a routine part of every pet’s annual wellness check and following the successful international rollout of our canine cancer test, we have now demonstrated that our technology is also effective for cats.
“We look forward to completing product development and making the Nu.Q® Vet Feline Test available through our distribution networks including reference laboratories and point-of-care platforms.
“The Nu.Q® Vet Canine test is already available in more than 20 countries, and we believe the addition of a feline equivalent could potentially double our total addressable market in the companion animal space.”
The Nu.Q® Vet Feline Test taps into a significant, rapidly growing market:
- Large Population: There are approximately 73.8 million pet cats in the United States alone6.
- High Risk: It is estimated that approximately 1 in 5 cats will develop cancer in their lifetime7. Feline lymphoma, the most common cancer in the species, is a primary target for the Nu.Q® assay.
- The “Senior” Segment: Over 25% of U.S. cats are considered seniors (aged 8+)8, representing a recurring annual screening opportunity for over 18 million pets in the U.S.6,8
- Market Growth: The North American veterinary oncology market is projected to reach $1.77 billion by 2025, with the feline segment expected to grow at a faster Compound Annual Growth Rate than the canine segment due to increasing owner awareness and a growing focus on feline-specific treatments”9.
The paper should be available on a preprint service in the coming days.
- Data on File
- Vail D, Thamm D, Liptak J, eds. Withrow and MacEwen’s Small Animal Clinical Oncology. 6th ed. Elsevier Health Sciences; 2019.
- M. Wilson‑Robles et al, BMC Vet Res, 2022, https://doi.org/10.1186/s12917-022-03429-8
- Dolan et al, BMC Vet Res, 2021 https://doi.org/10.1186/s12917-021-02991-x
- M. Wilson‑Robles et al, BMC Vet Res, 2021, https://doi.org/10.1186/s12917-021-02934-6
- AVMA Pet Ownership Sourcebook (2025) & APPA National Pet Owners Survey (2025)
- Can Cats Get Cancer? Understanding Feline Cancers Like Lymphoma & SCC. Imprimed
- Data on file, Volition TAM model
- Towards Healthcare: Veterinary Oncology Market Insights (October 2025)
###
- Share
- Tweet
- Share on Facebook
- Share
Subscribe for Volition product updates.
Or, click here for Investor Updates and press releases.
"*" indicates required fields
